An in-depth analysis approach enabling precision single chain nanoparticle design†
Abstract
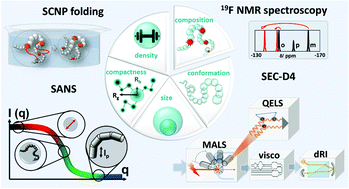
The synthesis of single chain nanoparticles (SCNPs) is a vibrant field in macromolecular science, enabled by a rich variety of synthetic strategies to induce macromolecular chain folding. Due to the decrease of the hydrodynamic volume upon folding, SCNP formation is typically characterized by a shift towards higher elution volumes in size exclusion chromatography (SEC). However, a step-change in the methodologies for SCNP analysis is required for the in-depth understanding of the nature of intramolecular polymer folding and internal SCNP structure, which is critical to enable their application as catalytic nanoreactors. Herein, we exploit a unique combination of small-angle neutron scattering (SANS), 19F NMR spectroscopy, and quadruple detection SEC to generate an encompassing and systematic view of the folded morphology of poly(tert-butyl acrylate) based-SCNPs as a function of their reactive group density (5, 15, and 30 mol%) and absolute molar mass (20, 50, 100 kDa). In addition to detailed morphological insights, we establish that the primary factor dictating the compaction of SCNPs is their reactive group density, which is ineffective below 5 mol%, reaching maximum compaction close to 30 mol%. The molar mass of the precursor polymers has a minor impact on how an SCNP compacts for molar masses above 20 kDa.


 Please wait while we load your content...
Please wait while we load your content...