Facile synthesis of a porous polynorbornene with an azobenzene subunit: selective adsorption of 4-nitrophenol over 4-aminophenol in water†
Abstract
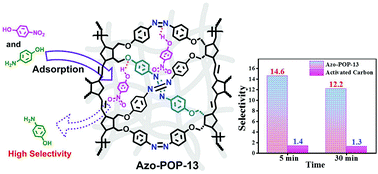
This paper reports the design and facile synthesis of porous polynorbornenes containing a 4,4′-dialkoxy-azobenzene subunit (named Azo-POP-13 and Azo-POP-14). They were synthesized by the robust reductive azo-coupling polymerization of the Ring-Opening-Metathesis-Polymerization (ROMP) product on bis-para-nitrophenylether of the norbornene diol. The polymerization rate of exo-NBNPE (kapp = 18.7 × 10−2 s−1) is faster than that of endo-NBNPE (kapp = 3.64 × 10−2 s−1). The as-synthesized Azo-POP-13 was found to selectively adsorb 4-nitrophenol (4-NP) over 4-aminophenol (4-AP) in water, with a selectivity up to 14.6 and good recyclability.


 Please wait while we load your content...
Please wait while we load your content...