A monocomponent bifunctional benzophenone–carbazole type II photoinitiator for LED photoinitiating systems†
Abstract
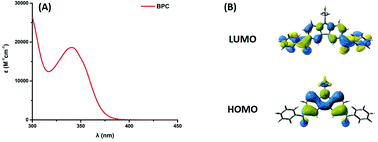
A bifunctional benzophenone–carbazole-based photoinitiator (BPC) was designed from its molecular structure viewpoint. To achieve this, two benzoyl substituents were introduced as peripheral groups of carbazole, so that a molecule in which two benzophenones were fused to one carbazole unit could be obtained in a one-step procedure. As a result of this combination, synergetic effects are thus achieved. Interestingly, BPC could efficiently initiate the free radical photopolymerization (FRP) of acrylates without addition of any extra hydrogen donors, demonstrating that not only a monocomponent Type II behavior but also fast photoinitiation rates were obtained in the presence of an amine and/or an iodonium salt under LED@365 nm irradiation. Meanwhile, BPC can also interact with the iodonium salt to generate cationic species for the cationic photopolymerization (CP) of epoxides. BPC was found as a versatile photoinitiator that can be used for both radical and cationic polymerizations. The best photoinitiating system (PIS) determined during the FRP tests was selected for 3D printing experiments. Steady state photolysis and fluorescence quenching experiments were carried out to verify the interaction between BPC and the different additives in the excited state. Electron spin resonance-spin trapping experiments successfully distinguished the free radicals generated by the different PISs and the proposed electron transfer mechanisms are also discussed.


 Please wait while we load your content...
Please wait while we load your content...