Comprehensive studies of the ligand electronic effect on unsymmetrical α-diimine nickel(ii) promoted ethylene (co)polymerizations†
Abstract
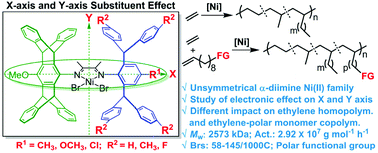
The ligand electronic effect plays a significant role in tuning the catalytic activity, molecular weight and topology of polymers, and comonomer incorporation in ethylene (co)polymerization; however, studies are rather limited in the milestone α-diimine late transition metal catalysts. In this contribution, by tailoring a sterically encumbered pentiptycenyl/dibenzhydryl substituted framework, the ligand electronic effects derived from both the para-position of the N-aryl group (horizontal axis: Me, MeO, and Cl) and the para-position of the dibenzhydryl moiety (vertical axis: Me, H, and F) are comprehensively investigated in unsymmetrical α-diimine Ni(II) promoted ethylene (co)polymerizations for the first time. In the ethylene polymerization, the electron-withdrawing Cl group (horizontal axis) prefers to give a higher branching density (145/1000 C) with higher catalytic activity (29 200 kg mol−1 h−1), while the electron-donating Me group affords a higher molecular weight (2573 kDa). Moreover, the electron-withdrawing F group (vertical axis) again generates a higher branching density, but a lower molecular weight with reduced catalytic activity. In contrast, in the ethylene copolymerization with methyl 10-undecenoate, the electron-donating Me group derived from both the horizontal axis and vertical axis is concurrently beneficial, giving an increased polymer molecular weight (374 kDa) and comonomer incorporation with higher catalytic activity. However, all of the electron-withdrawing groups coming from either the horizontal axis (Cl) or vertical axis (F) is not good for copolymerization. This work sheds light on the different effects of electronic substituents on ethylene polymerization and ethylene-polar monomer copolymerization.


 Please wait while we load your content...
Please wait while we load your content...