Structural insight into the viscoelastic behaviour of elastomeric polyesters: effect of the nature of fatty acid side chains and the degree of unsaturation†
Abstract
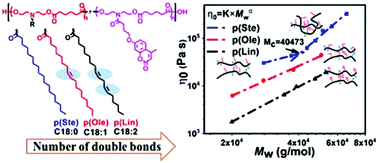
Degradable polymers derived from sustainable sources, such as vegetable oils, provide an eco-friendly alternative to petroleum-based polymers. Vegetable oils are composed of triglycerides which differ in their fatty acid chain length and the degree of unsaturation. However, vegetable oils can vary greatly in their composition depending on the type of oil and region of cultivation, and hence, polymers made from such oils can result in heterogeneous polymer properties. Therefore, herein we synthesized three polyesters with different pendant fatty acid groups (linoleic acid C18:2, oleic acid C18:1, and stearic acid C18:0) and evaluated the effect of the structural difference on the rheological and mechanical properties of these vegetable oil based polyesters. Increasing the degree of unsaturation of the fatty acid side chains decreased the glass transition temperature, zero-shear viscosity, tensile strength, elongation-at-break, and the recovery efficiency of elastic energy (resilience) of its crosslinked elastomer. We hypothesize that the cis double bonds introduce ‘kinks’ to the fatty acid side chains that impede the packing of the polymer chains and therefore, act as efficient ‘internal diluents’. This is corroborated by an increase in the critical molecular weight for entanglement of the polymers with increasing number of cis double bonds in the side chains. Moreover, the presence of cis double bonds was associated with an increase in intermolecular friction and a decrease in the resilience. The structural insights derived from this study will be useful to design low modulus polymers and predict the processing and mechanical properties of polymers with fatty acid side chains.


 Please wait while we load your content...
Please wait while we load your content...