Using the dynamic behavior of macrocyclic monomers with a bis(hindered amino)disulfide linker for the preparation of end-functionalized polymers†
Abstract
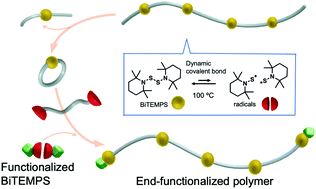
Polymer topology transformations based on dynamic covalent chemistry (DCC) have attracted broad interest as the macroscopic properties of polymers can be freely changed in response to the polymer topology even after their synthesis. However, controlling the primary structures that accompany the topology transformation via DCC remains challenging. Herein, a method is presented that allows controlling functional end groups and molecular weight in DCC-based polymers using a combination of macrocyclic monomers and bifunctional acyclic compounds that contain bis(2,2,6,6-tetramethylpiperidin-1-yl)disulfide (BiTEMPS) moieties. The BiTEMPS moieties are stable at room temperature, but exchange disulfide bonds above 80 °C. Specifically, end-functionalized polymers were synthesized by simply heating a mixture of a macrocyclic compound with one BiTEMPS moiety and bifunctional acyclic BiTEMPS compounds as sources of repeat units and terminal groups, respectively. In the present method, various end-functionalized polymers including A–B–A type triblock copolymers with roughly controlled molecular weight were prepared by changing the chemical structures of the bifunctional BiTEMPS compounds and their feed ratio relative to the cyclic monomers.


 Please wait while we load your content...
Please wait while we load your content...