Turning natural δ-lactones to thermodynamically stable polymers with triggered recyclability†
Abstract
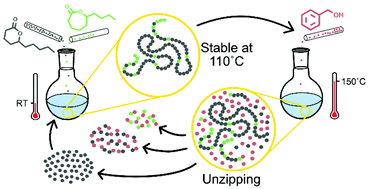
To extend the use of naturally occurring substituted δ-lactones within the polymer field, their commonly low ceiling temperature and thereby challenging equilibrium behavior needs to be addressed. A synthetic strategy to control the polymerization thermodynamics was therefore developed. This was achieved by copolymerizing δ-decalactone (δDL) with either ε-decalactone (εDL) or ε-caprolactone (εCL) at room temperature (RT), with diphenyl phosphate (DPP) as catalyst. The thermodynamic stability of PδDL-co-εDL and PδDL-co-εCL increased with increased comonomer ratio in the feed, to 10% and 30% monomeric δDL, respectively, at 110 °C. This is in contrast to the PδDL homopolymer, which under the same conditions depolymerized to 70% monomeric δDL at equilibrium. The copolymers’ macromolecular structure, originating from the copolymerization kinetics, was found to be the crucial factor to mitigate δDLs equilibrium behavior. To close the loop, designing materials for a circular economy, the recycling of PδDL-co-εDL was demonstrated, by reaction with benzyl alcohol (BnOH) as an external nucleophile, leading to cyclic monomers or dimers with BnOH at high yield.
- This article is part of the themed collection: Plastics in a circular economy


 Please wait while we load your content...
Please wait while we load your content...