Vesicular assemblies of thermoresponsive amphiphilic polypeptide copolymers for guest encapsulation and release†
Abstract
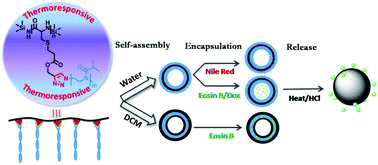
Synthetic polypeptides are bio-inspired materials with good self-assembling capabilities. However, their hydrophobic nature restricts their use in biomedical applications. Pseudopeptidic poly(2-oxazoline)s are also bio-inspired and water-soluble polymers. Thus, the motivation for this research work is to integrate these two units to produce amphiphilic copolymers with a high propensity to self-assemble into vesicular nanostructures that are useful for various potential applications. To achieve this, a convenient “click” grafting technique is utilized to prepare water-soluble amphiphilic poly(cysteine)-graft-poly(2-isopropyl-2-oxazoline) (PCys-g-PiPOx) copolymers. This involves synthesis of propargylated polycysteine (PCys-S-Pr) and azide-functionalized poly(2-isopropyl-2-oxazoline) (PiPOx-N3) blocks via ring-opening polymerization (ROP) of the corresponding N-carboxyanhydride (NCA) and cationic ring-opening polymerization (CROP) techniques, respectively, followed by their grafting. The grafting of the PiPOx block introduces thermoresponsiveness in the copolymer, exhibiting LCST phase behaviour in water, whose cloud point can be tuned by varying the copolymer's composition and concentration. The amphiphilic nature of PCys-g-PiPOx molecules drives them to self-assemble into nanostructures with vesicular morphology in aqueous and nonaqueous media, as clearly evident from the transmission electron microscopic images. These polypeptide copolymer vesicles are capable of encapsulating both hydrophobic and hydrophilic dyes present in different environments and confirmed through fluorescence and confocal microscopy. These copolymer vesicles can also be highly efficiently used for encapsulating the drug doxorubicin and their subsequent triggered release was also studied by absorption and emission spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...