Functionalization of amine-cured epoxy resins by boronic acids based on dynamic dioxazaborocane formation†
Abstract
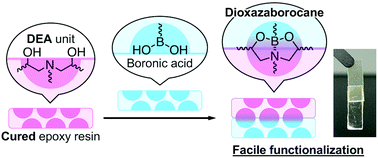
Functionalization of epoxy resins after curing is challenging because of their thermosetting properties. The intrinsic reactive chemical structures of epoxy resins can be used to overcome this challenge. Herein, we demonstrated functionalization of post-cure epoxy resins based on dynamic dioxazaborocane (DOAB) formation between diethanolamine (DEA) units in amine-cured epoxy resins and boronic acid modifiers. The cured epoxy resins were modified using a phenylboronic acid and a linear polymer with boronic acid pendants functionalized with spiropyran groups as a dye and a functional coating. Furthermore, cross-linked polymers containing boronic acid groups, i.e., an acrylamide gel and a methacrylic elastomer, were synthesized, which behaved as adhesives for cured epoxy resins. These results provide novel possibilities for application of dynamic DOAB formation to functionalize amine-cured epoxy resins.
- This article is part of the themed collection: Chemistry for Covalent Adaptable Polymer Networks


 Please wait while we load your content...
Please wait while we load your content...