Epoxy vitrimers incorporating physical crosslinks produced by self-association of alkyl chains†
Abstract
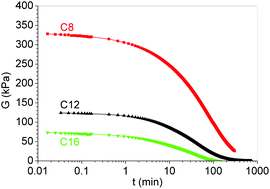
Epoxy vitrimers were synthesized employing stoichiometric amounts of diglycidyl ether of bisphenol A (DGEBA), sebacic acid (20% eq.) and an n-alkylamine, either octylamine (OA), dodecylamine (DA) or hexadecylamine (HA) (80% eq.). Apart from the chemical crosslinks generated by diester groups formed in transesterification reactions, these vitrimers include physical crosslinks arising from self-association of alkyl chains. The relative fraction and strength of physical crosslinks was high for octylamine (OA), intermediate for dodecylamine (DA) and low for hexadecylamine (HA). This was reflected by rubbery moduli that increased significantly from HA to OA. The covalently bonded tertiary amine generated in the epoxy-amine addition reaction was an efficient catalyst of transesterification reactions. Relatively fast stress relaxation rates were observed driven by transesterification reactions coupled to the dynamic exchange of self-associated alkyl chains. The occurrence of this last mechanism was evidenced by the stress relaxation observed for the linear polymer synthesized with stoichiometric amounts of DGEBA and OA that forms a permanent network up to temperatures close to degradation due to the presence of a high fraction of self-associated n-octyl chains. These vitrimers might find applications in several fields such as soft robotics, electro and magnetorheological elastomers, soft tissues and in additive manufacturing.
- This article is part of the themed collection: Chemistry for Covalent Adaptable Polymer Networks


 Please wait while we load your content...
Please wait while we load your content...