Chiral expression of co-crystallizing poly(thiophene)-block-poly(selenophene) copolymers†
Abstract
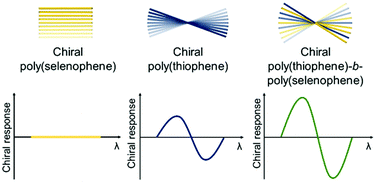
In this report, the chiral expression of co-crystallizing conjugated poly(thiophene)-b-poly(selenophene) copolymers is investigated. For this purpose, different poly(thiophene)-b-poly(selenophene) copolymers were synthesized via controlled Kumada catalyst transfer condensative polymerization (KCTCP). The homopolymers, poly(thiophene) and poly(selenophene), are synthesized as a reference. Solvatochromism experiments were conducted to investigate the (chiral) self-assembly of all the polymers using UV-vis and circular dichroism (CD) spectroscopy. It was found that, although the chiral homopoly(selenophene) surprisingly did not self-assemble in a chiral way, the block copolymers did show a chiral expression. This can only be explained by the suggested co-crystallization of the block copolymers, since for (micro)phase-separating block copolymers no chiral expression would be expected, as the first aggregating block, in this case the poly(selenophene) block, then would dictate its stacking to the second block. Differential scanning calorimetry (DSC) further suggested the co-crystallization of the block copolymers. Moreover, the chiral expression of the poly(thiophene)-b-poly(selenophene) copolymers was even larger than for the homopoly(thiophene), which can be explained by the increased disorder present in the block copolymers. Dynamic light scattering (DLS) eliminated the existence of a correlation between the chiral response and the size of the polymer aggregates, further evidencing that the increase in the chiral response of the block copolymers is solely due to the suggested co-crystallization of the block copolymers.


 Please wait while we load your content...
Please wait while we load your content...