Surface-initiated polymerization for the preparation of magnetic polymer composites†
Abstract
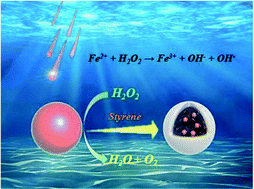
In organic–inorganic hybrid materials, it is difficult to obtain magnetic polymer microspheres with the good encapsulation of magnetic materials because there are few successful interactions between monomers and magnetic materials. In this study, a method involving surface-initiated polymerization was developed for the preparation of magnetic polymer composites; the method used Fe3O4 nanoparticles and H2O2 as a redox initiator. After activation with carboxylic acid, the amount of Fe2+/Fe3+ on the surfaces of the Fe3O4 nanoparticles increases, which can effectively catalyze the decomposition of H2O2 to produce hydroxyl radicals. The hydroxyl radicals were used to initiate the polymerization of the monomers on the surfaces of the Fe3O4 nanoparticles so that the Fe3O4 nanoparticles became encapsulated in polymer microspheres and in this way highly magnetic responsive polymer microspheres were obtained. This method was found to be applicable to the polymerization of many types of monomers, and some polymer microspheres (PS, PMMA, PBA and PVAc) were synthesized successfully. The effects of reaction time, concentration of the added Fe3O4, volume of the added styrene monomer, and volume of the added H2O2 on the prepared Fe3O4@PS composites were investigated. The results indicated that the polymer microsphere size was controllable (60 nm–2 μm) and the load of Fe3O4 nanoparticles in the polymer microspheres could be adjusted by the addition of Fe3O4 nanoparticles. This method was beneficial for preparing various magnetic polymer composites having significant potential application value in many fields.


 Please wait while we load your content...
Please wait while we load your content...