Fluorinated vesicles embedded with Ru-based catalysts as efficient and recyclable nanoreactors for photo-mediated aerobic oxidation†
Abstract
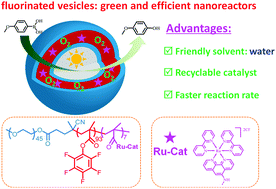
From the point of view of green chemistry, it is extremely desirable that organic chemistry works efficiently in an environmentally friendly way. Recyclable vesicles having a fluorinated membrane with embedded Ru-based catalysts were generated via self-assembly of a PEG-b-PPFMA/Ru(bpy)2(phen-NH2)Cl2 amphiphilic copolymer (PPFMA = poly(pentafluorophenyl methacrylate), bpy = bipyridyl and phen-NH2 = 5-amine-1,10-phenanthroline), which was synthesized via RAFT polymerization of a PFMA monomer using a PEG-based chain transfer agent, followed by the incorporation of an Ru-based catalyst via amidation between the PPFMA block and Ru(bpy)2(phen-NH2)Cl2. These fluorinated vesicles can serve as nanoreactors for aerobic oxidative hydroxylation of arylboronic acid. Owing to the presence of these nanoreactors, this reaction proceeded smoothly and efficiently in aqueous media with an enhanced reaction rate using less catalyst as compared to previous work. Significantly, the nanoreactors could be recycled via ultrafiltration and reused for at least five cycles with a slight decrease in the yield. Moreover, it is found that the fluorinated vesicles showed an enhanced reaction rate in the photo-mediated aerobic oxidative hydroxylation of arylboronic acid compared to the nanoreactors of vesicles containing the same content of the Ru-based catalyst with a similar size and morphology, but with a non-fluorinated membrane. To the best of our knowledge, this is the first example concerning a fluorinated nanoreactor for efficient aerobic oxidation by taking advantage of the enhanced oxygen solubility and diffusivity of fluorinated domains. Due to the high reactivity of pentafluorophenyl ester toward amino-based compounds and excellent functionality tolerance of RAFT polymerization, the strategy described provides a versatile platform to prepare efficient nanoreactors containing a variety of catalysts for diverse reactions, especially for aerobic oxidation.


 Please wait while we load your content...
Please wait while we load your content...