RAFT coupling chemistry: a general approach for post-functionalizing molecularly imprinted polymers synthesized by radical polymerization†
Abstract
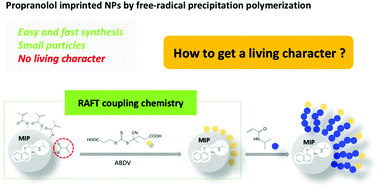
Molecularly imprinted polymers (MIPs) are well-known as antibody mimics, with binding properties for their target on a par with those of biomacromolecules such as antibodies or enzymes. A great majority of MIPs is nowadays synthesized by free-radical polymerization (FRP), owing to the mild reaction conditions, tolerance for monomer and solvent impurities and high flexibility in terms of polymerization setups. Nevertheless, FRP MIPs lack suitable functionalities for a further elaboration of their structures via, for instance, extension with consecutive blocks. This results in practical limitations for FRP imprinted scaffolds in terms of adapting them to a given application. In this work, we show that FRP MIPs can easily be functionalized with “living” moieties for chain extension, by coupling a chain transfer (RAFT) agent to the ubiquitous unreacted double bonds on the MIP surface. Imprinted nanoparticles synthesized by precipitation polymerization, as well as microparticles obtained by bulk polymerization and mechanical milling, were reacted with a dithioester or a trithiocarbonate in the presence of a radical source. This resulted in RAFT coupling which allowed the surface grafting of polymer brushes, while the imprinted scaffolds retained their binding properties in terms of template affinity and selectivity. Upon extension with poly(N-isopropylacrylamide) p(NIPAm), RAFT-coupled MIPs also acquired excellent binding properties in aqueous media, compared to unmodified particles which just exhibited non-specific binding. Thus, this easy approach, which takes advantage of a common feature of cross-linked particles obtained by FRP, is expected to be of general interest due to the easy post-modification and fine-tuning of the material's surface properties.


 Please wait while we load your content...
Please wait while we load your content...