Thermally healable and reprocessable polymethacrylate networks based on diol-mediated metathesis of 6-membered boronic esters†
Abstract
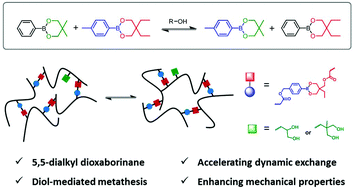
Finely tuning the exchange rate of a dynamic motif is important to manipulate the properties of vitrimers or covalent adaptable networks. Herein, we report a new type of thermally repairable and reprocessable poly(butyl methacrylate) (PBMA) network crosslinked by a dynamic six-membered phenylboronic ester linkage. First, we studied the metathesis kinetics of small molecule 5,5-dialkyl-2-phenyldioxaborinanes with or without a catalytic amount of diols or mono-alcohol. These alcohols could drastically decrease the activation energy of the metathesis reaction through a mechanism of hydroxy-assisted ring opening process. Next, two PBMA copolymers containing complementary boronic esters were prepared, and the heat-induced sol–gel–sol transformation of a mixture of the two copolymers in anisole was confirmed through dynamic crosslinking. Subsequently, PBMA networks with different crosslinking densities were prepared by redox initiated radical polymerization, using a dioxaborinane-containing dimethacrylate. These bulk networks showed significant stress relaxation upon heating to 100 °C and could be completely healable or reprocessable at 150 °C. In contrast, they possessed good creep resistance at 50 °C, a temperature that was around the Tg of the networks. Finally, we incorporated different diol units into the PBMA networks and investigated the effects of the structure and content of the diols on the thermal and mechanical properties, rheological behaviors, and thermal healing efficiency of the materials. The pendant diols could effectively reduce the healing/reprocessing temperature of the networks by accelerating metathesis of the boronic ester linkages. Meanwhile, the mechanical strength of the networks was significantly enhanced by these diol units due to the formation of hydrogen bonds.


 Please wait while we load your content...
Please wait while we load your content...