Zwitterionic copolymerization of γ-butyrolactone with 3,3-bis(chloromethyl) oxacyclobutane catalyzed by scandium triflates†
Abstract
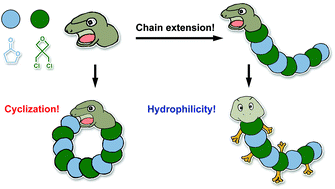
Bioderived poly(γ-butyrolactone) (PBL) is a biodegradable and biocompatible material with properties superior to other aliphatic polyesters. Ring-opening polymerization (ROP) of γ-butyrolactone (BL) remains a great challenge due to the non-strained structure and unfavorable thermodynamics. In the present work, we design a novel zwitterionic ROP system for a well-controlled cationic copolymerization of BL with 3,3-bis(chloromethyl) oxacyclobutane (CO) catalyzed by scandium triflates (Sc(OTf)3). The molar fraction of BL repeating units in the random copolymer keeps 50% which is independent of the monomer conversions or polymerization time when the feed ratio of [BL]/[CO] is higher than 2. High molecular weight (MW) linear and cyclic copolymers of poly(BL-co-CO) are synthesized by inter- and intra-molecular coupling reactions, respectively, after quantitative consumption of CO. Cyclic copolymers possess higher decomposition and glass transition temperatures than the linear ones with comparable MWs due to the absence of terminal groups. Chloromethyl groups of CO repeating units provide PBL with reactive sites for further functionalization. A post-polymerization quaternization of N-methylimidazole produces amphiphilic copolymers able to self-assemble into nano-aggregates in aqueous solution.


 Please wait while we load your content...
Please wait while we load your content...