Supramolecularly cross-linked amphiphilic block copolymer assembly by the dipolar interaction of a merocyanine dye†
Abstract
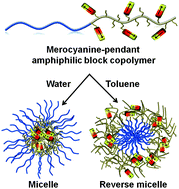
The ability of amphiphilic block copolymers to produce a wide range of self-assembled structures from nano to mesoscale makes them potentially relevant candidates in biomedical research and in materials applications. Hence, a fundamental understanding of their self-assembly behavior is of utmost importance. In this article, we have presented a novel supramolecular approach based on dipole–dipole interactions of one class of merocyanine (MC) dyes for maneuvering remarkably stable micelles with non-covalently cross-linked core and shell in water and toluene, respectively, from a PEG-b-polystyrene based amphiphilic block copolymer (MC-P1) with statistically distributed MC dyes within the hydrophobic polystyrene block. We have elucidated various physical properties of the amphiphilic block copolymer assembly in both water and toluene. Astonishingly, high thermal stability, extremely slow dynamics of mixing of the micelles as is evident from Förster resonance energy transfer (FRET) experiments and a very low critical micelle concentration were attributed to the hydrophobically assisted dipole–dipole interaction mediated supramolecular dimerization of pendant MCs. Analogous physical properties observed in the two tested solvents of contrastingly different solvent polarities illustrate similar packing behavior of the MC dyes within the micelles and the reverse micelles. Unlike the conventional strategies for covalent cross-linking, this novel supramolecular cross-linking approach by MC dimerization bestows additional features such as rich optical properties and dimerization-induced enhanced emission, besides reinforcing the polymeric micelles. Considering the impact of merocyanines in photovoltaics and non-linear optics, their conjugation to the polymeric domain is also anticipated to bring about a new class of MC-appended polymer assemblies for potential organic materials.
- This article is part of the themed collection: Chemistry of polymers - Chemical Science symposium collection


 Please wait while we load your content...
Please wait while we load your content...