AIE-active polysulfates via a sulfur(vi) fluoride exchange (SuFEx) click reaction and investigation of their two-photon fluorescence and cyanide detection in water and in living cells†
Abstract
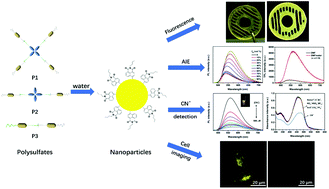
Three polysulfates P1, P2 and P3 containing two representative AIE-active groups, tetraphenyl ethylene and naphthylamide, were successfully synthesized based on a sulfur(VI) fluoride exchange (SuFEx) click reaction. Their solid fluorescence was studied in detail and liquid fluorescence emission could be tuned by choosing solvents with different polarities. P1 with a branched structure showed higher fluorescence emission than P2 and P3 both in the solid state and in organic solutions because of the more crowded local environment of the tetraphenyl ethylene group in the main chain. Moreover, all the polymers exhibited typical aggregation induced emission (AIE) effects in the solid state and in DMF/water solutions. Furthermore, their two-photon fluorescence behaviours were similar to their one-photon fluorescence behaviours in different organic solvents and in 90% DMF/water solutions. Interestingly, they could selectively detect cyanide ions with lower detection limits in water on account of the anion–π interactions between the naphthylamide groups and the cyanide ions. The detection limits of P1 (0.137 μM) and P2 (0.131 μM) are lower than P3 (0.145 μM), which may be attributed to the more exposed active sites of the P1 and P2 nanoparticles (NPs). All the results indicate that these polymers have higher sensitivity and better applicability for cyanide ion detection in water and in living cells.


 Please wait while we load your content...
Please wait while we load your content...