Real-time near-infrared fluorescence reporting the azoreductase-triggered drug release†
Abstract
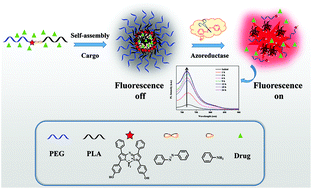
A near-infrared fluorescent probe plays an important role in biosensing and bioimaging because of its high efficiency, sensitivity and negligible background interference in vivo. Azobenzene derivatives demonstrate well-known sensitivity to light and are unique enzyme-responsive candidates in drug delivery and biological detection because the azo double bond can be cleaved by azoreductase. Herein, a novel near-infrared probe of an amphiphilic block copolymer with biological compatibility was synthesized. The azobenzene group attached with the near-infrared fluorescent group AzaBODIPY (boron-dipyrromethenes) linked the hydrophilic (PEG) and hydrophobic (PLA) segments to form the amphiphilic block copolymer PEG-AzaBODIPY-AZO-PLA. PEG398-AzaBODIPY-AZO-PLA144 self-assembled into drug-loaded spherical micelles. The self-assemblies were non-fluorescent owing to the aggregation-induced quenching (ACQ) effect. Triggered by azoreductase, the PEG and PLA segments were disconnected due to the cleavage of the azo bond. This caused the disassembly of the micelles and the subsequent release of the encapsulated drug. Upon disassembly, the emission of the near-infrared fluorescence was activated by the elimination of the ACQ effect. Furthermore, the fluorescence intensity of the self-assemblies in solution increased continuously with drug release. The fluorescence reporting drug release demonstrates the potential applications in biosensing and controlled drug release in the colon of the human intestine.


 Please wait while we load your content...
Please wait while we load your content...