Conjugated electrochromic polymers with amide-containing side chains enabling aqueous electrolyte compatibility†
Abstract
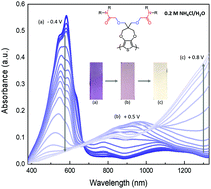
Conjugated polymers based on poly(3,4-propylenedioxythiophene) (PProDOT) backbones are widely studied for electrochromics, due to their favourable redox properties including enhanced optical contrast and fast switching kinetics in nonaqueous electrolytes. The usual hydrophobic nature of these polymers renders them redox-inactive when interfaced with aqueous salt solutions. This work illustrates an effective side chain modification approach where polar amide functional groups are incorporated into the polymer side chains to attain redox switching in aqueous electrolytes while maintaining a high contrast. The impact of substitutional modifications (in terms of primary and secondary amide groups) on the interaction with hydrated and non-hydrated ions is studied using electrochemical and spectroscopic techniques to elucidate the underlying mechanisms leading to redox activity in aqueous electrolytes. The presence of amide groups is beneficial in terms of reducing the oxidation onset potential, making them attractive candidates for electrochromics in both nonaqueous and aqueous electrolytes.
- This article is part of the themed collection: Polymer Chemistry Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...