Non-solvating, side-chain polymer electrolytes as lithium single-ion conductors: synthesis and ion transport characterization†
Abstract
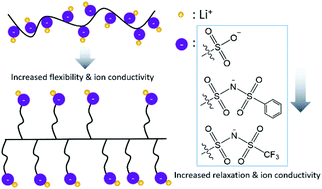
Solid-state single-ion conducting polymer electrolytes have drawn considerable interest for secondary lithium batteries due to their potential for high electrochemical stability and safety, but applications are limited by their low ionic conductivities. Specifically, poly(ethylene oxide) (PEO) based electrolytes have the highest reported Li+ conductivities for these materials; however, their potential is limited due to the ion transport mechanism being coupled to segmental relaxations of the cation solvating polymer chain. To investigate the potential of single-ion conducting polymer electrolytes lacking polar matrices, we synthesized three para-polyphenylene-based, side-chain polymer electrolytes with various pendent anion chemistries (–SO3−, –PSI−, and –TFSI−) with differing binding affinities to Li+. Compared with the previously reported lithium poly(4-styrenesulfonyl(trifluoromethylsulfonyl)imide) (LiPSTFSI), the side-chain polymers showed at least 3 orders of magnitude higher conductivity with the same –TFSI− anion (6.7 × 10−6 S cm−1 compared with 1.2 × 10−10 S cm−1 at 150 °C). We found that the side-chain electrolyte showed a dielectric relaxation dominated transport mechanism through use of dielectric spectroscopy analysis. The conductivity is highly dependent on the charge delocalization and size of the pendent anion, which provides a pathway forward for the engineering of polymeric ion conductors for electrochemical applications.
- This article is part of the themed collection: Polymer Chemistry Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...