Ligand–metal secondary interactions in phosphine–sulfonate palladium and nickel catalyzed ethylene (co)polymerization†
Abstract
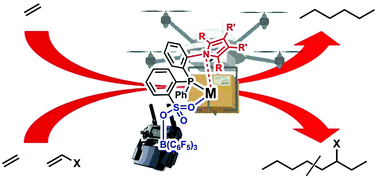
Ligand–metal secondary interactions are an efficient strategy to modulate transition-metal-catalyzed ethylene (co)polymerizations. In this contribution, some phosphine–sulfonate Pd and Ni catalysts, bearing sp2 hybrid N based aromatic carbazolyl and pyrrolyl units in their ligand framework, have been designed and synthesized. The N-heterocyclic unit can interact with the Pd or Ni center, thus inducing ligand–metal secondary interactions. Moreover, these secondary interactions could be modulated through the addition of a Lewis acid reagent. The Pd catalysts exhibit high activities in ethylene polymerization and copolymerization with a variety of polar comonomers (methyl acrylate, acrylic acid, 6-chloro-1-hexene, 10-undecenol and 10-undecenoic acid), resulting in high molecular weight (co)polymers. The Ni catalysts show low activities, affording low molecular weight (co)polymers. Both the Pd and Ni catalysts demonstrate enhanced thermal stability versus their analogues devoid of the ligand–metal interaction. The Lewis acidic additive B(C6F5)3 can efficiently increase activities and decrease polymer molecular weights. Specifically, a 670-fold increase of the chain transfer rate can be achieved in Pd catalyzed ethylene polymerization. The Lewis acid addition can also influence polymer microstructures in Ni catalyzed ethylene polymerization. The combination of ligand–metal secondary interactions with Lewis acid modulation makes this strategy highly versatile and potentially applicable to other catalytic processes.
- This article is part of the themed collections: Polymer Chemistry Most Popular 2020 and Polymer Chemistry Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...