Impact of metal cations on the thermal, mechanical, and rheological properties of telechelic sulfonated polyetherimides†
Abstract
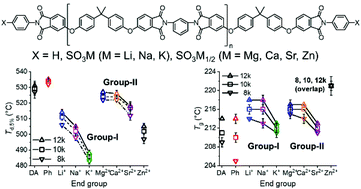
Herein we have synthesized telechelic sulfonated polyetherimides (PEIs) with a series of metal cations, including alkali metal cations (Li+, Na+, and K+) and alkaline earth metal cations (Mg2+, Ca2+, and Sr2+). By comparing the metal sulfonated PEIs with a noncharged analog phenyl-terminated PEI, we have systematically investigated how the metal cations affect the thermal, mechanical, and rheological properties of PEIs. PEIs with alkaline earth metal cations showed higher thermal degradation temperatures than those with alkali metal cations. All sulfonated PEIs showed lower thermal degradation temperatures than noncharged PEIs. All metal cations except K+ and Sr2+ increased the glass transition temperatures of sulfonated PEIs. Most metal (except K and Sr) sulfonated PEIs possessed good film formability when the molecular weight was above 10 kDa. Due to their large cation sizes, the K and Sr sulfonated PEIs exhibited reduced glass transition temperatures and inferior film formability. Alkali metal sulfonated PEIs showed high thermo-oxidative stability, but none of the alkaline earth metal sulfonated PEIs exhibited good thermo-oxidative stability. The rheological time–temperature superposition revealed that the interaction strength between the sulfonate anions and the alkali metal cations decreased as the cation size increased in the sequence of Li+, Na+, and K+. This study provides a guideline for synthesizing metal sulfonated PEIs and other metal sulfonated polymers with tailored thermal, mechanical, and rheological properties.
- This article is part of the themed collection: Polymer Chemistry Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...