Role of E270 in pH- and metal-sensitivities of firefly luciferases†
Abstract
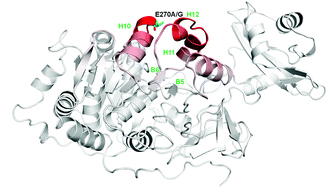
Firefly luciferases display a typical change in bioluminescence color to red at acidic pH, high temperatures and in the presence of heavy metals. Recently, the proton and metal sensing site responsible for the pH-sensitivity of firefly luciferases, which involves the salt bridges between E311-R337 and H310-E354, was identified. However, it is unclear what other residues contribute to the distinct degrees of pH-sensitivity observed in other firefly luciferases. A multialignment of primary structures of a large set of pH-sensitive and pH-insensitive beetle luciferases showed that the conserved E270 among adult firefly luciferases is substituted by Gly (railroad worms)/Gln (click-beetles) in pH-insensitive ones. Site-directed mutagenesis studies using Macrolampis sp2 and Amydetes vivianii firefly luciferases indeed showed that E270 is important for the pH-dependent activity and spectral profiles: the substitution E270A/G drastically decreases the spectral pH-sensitivity, and extends the activity profile above pH 9.0. These mutations also decrease the sensitivity to metals such as zinc, mercury and cadmium. Modelling studies showed that the residue E270 is located in a three-glutamate motif (269EEE271) at the N-terminal of α-helix-10. The results suggest that at acidic pH, the protonation of E270 carboxylate may extend a turn of the helix at the N-terminal, misaligning the pH-sensor and luciferin phenolate binding site residues: S286, I288 and E311. In contrast, the substitution of E270A/G may unwind a turn of the α-helix-10, indirectly increasing the interaction of the pH-sensor and other residues at the bottom of the luciferin binding site, stabilizing the green light emitting conformation.


 Please wait while we load your content...
Please wait while we load your content...