Composition, phosphorylation and dynamic organization of photosynthetic protein complexes in plant thylakoid membrane
Abstract
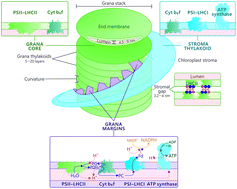
The photosystems (PS), catalyzing the photosynthetic reactions of higher plants, are unevenly distributed in the thylakoid membrane: PSII, together with its light harvesting complex (LHC)II, is enriched in the appressed grana stacks, while PSI–LHCI resides in the non-appressed stroma thylakoids, which wind around the grana stacks. The two photosystems interact in a third membrane domain, the grana margins, which connect the grana and stroma thylakoids and allow the loosely bound LHCII to serve as an additional antenna for PSI. The light harvesting is balanced by reversible phosphorylation of LHCII proteins. Nevertheless, light energy also damages PSII and the repair process is regulated by reversible phosphorylation of PSII core proteins. Here, we discuss the detailed composition and organization of PSII–LHCII and PSI–LHCI (super)complexes in the thylakoid membrane of angiosperm chloroplasts and address the role of thylakoid protein phosphorylation in dynamics of the entire protein complex network of the photosynthetic membrane. Finally, we scrutinize the phosphorylation-dependent dynamics of the protein complexes in context of thylakoid ultrastructure and present a model on the reorganization of the entire thylakoid network in response to changes in thylakoid protein phosphorylation.
- This article is part of the themed collections: 2020 Perspective article collection and The World Congress on Light and Life, Barcelona 2019


 Please wait while we load your content...
Please wait while we load your content...