Effect of ionic liquid on the fluorescence of an intramolecular exciplex forming probe†
Abstract
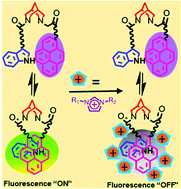
The effects of ionic liquid addition on the spectroscopic properties of a pyrene-tryptophan-containing fluorescent intramolecular complex in polar-aprotic and polar-protic solvents, specifically, acetonitrile and ethanol, are assessed. Two ionic liquid sets, consisting of seven different ionic liquids, were explored; set 1 comprised three imidazolium-containing ionic liquids paired with different anions while set 2 consisted of varying cations, namely, imidazolium, pyrrolidinium, ammonium, and pyridinium, partnered with a common anion, bis(trifluoromethylsulfonyl)imide ([Tf2N−]). The results provided herein reveal that all ionic liquids explored behave as quenchers, however, the imidazolium-, pyrrolidinium-, and ammonium-containing ionic liquids selectively quenched the fluorescence from the exciplex while the monomer emission from pyrene was largely unaffected relative to exciplex emission. Conversely, the pyridinium ionic liquid, significantly quenched the fluorescence from both the pyrene monomer and the pyrene-tryptophan exciplex, as was expected. The observed quenching is demonstrated to originate from the cations of the ionic liquids and is, in general, more efficient for an imidazolium ionic liquid that contains an acidic proton in the C2 position. Stern–Volmer plots of the exciplex quenching demonstrate a complex quenching mechanism that does not appear to follow any conventional quenching models with the data best fit to an exponential equation. Furthermore, time-resolved fluorescence measurements reveal that the quenching is not dynamic in nature as the recovered decay times do not systematically decrease with increasing ionic liquid concentration, suggesting a possible static quenching mechanism. Thus, the formation of a “dark” ensemble is proposed, in which the ionic liquid cations complex with or crowd around the exciplex, quenching the intramolecular energy transfer.


 Please wait while we load your content...
Please wait while we load your content...