Intramolecular interaction of synthetic chlorophyll heterodyads with different π-skeletons†
Abstract
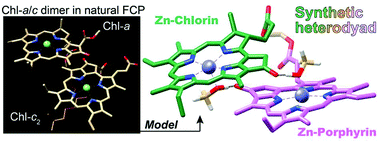
Two heterodyads were prepared from the chemical modification of naturally occurring (bacterio)chlorophyll-a and composed of a chlorin π-skeleton linked to a porphyrin or bacteriochlorin π-system. Zinc methyl pyropheophorbide-a, one of the chlorophyll-a derivatives, was covalently linked with its 17,18-didehydrogenated species (zinc methyl pyroprotopheophorbide-a) or its trans-7,8-dihydrogenated analog (zinc methyl pyrobacteriopheophorbide-a as one of the bacteriochlorophyll-a derivatives) through ethylene glycol diester at their 17-propionate residues. In benzene, the central zinc atoms of the synthetic conjugates were coordinated by two methanol molecules which were hydrogen-bonded with the 13-keto-carbonyl groups in a dyad molecule. The methanol locked, y-axis aligned, and slipped cofacial conformers showed two apparent Qy bands at longer wavelengths than those of the composite zinc complex monomers. The red-shifted Qy bands are ascribable to the exciton coupling of the two different π-systems in the folded heterodyad conformers. The synthetic heterodyads could be models of chlorophyll-a/c dimers in the light-harvesting antennas of chromophytes including fucoxanthin–chlorophyll proteins in diatoms and also chlorophyll-a species interacting with bacteriochlorophyll-a or g species in the charge-separating reaction centers of green sulfur bacteria or heliobacteria, respectively.


 Please wait while we load your content...
Please wait while we load your content...