Influence of a 4′-substituent on the efficiency of flavonol-based fluorescent indicators of β-glycosidase activity†
Abstract
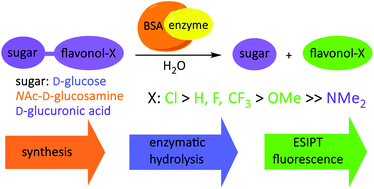
This article presents novel fluorescent probes, based on the excited-state intramolecular proton transfer (ESIPT) phenomenon and flavonols, sensitive to the action of specific glycosidases. 4′-Substituted flavonols were synthesized, using various approaches, and glycosylated with D-glucose, N-acetyl-D-glucosamine and D-glucuronic acid. Evaluation of the β-glycosidase activities was performed in neutral and acidic pH. In all the cases examined, an acidic environment accelerated enzymatic hydrolysis. It was demonstrated that the 4′-chloroflavonyl glycosides of all sugars tested, both in neutral and acidic pH, are the ones most sensitive to the presence of hydrolase. In turn, 4′-dimethylaminoflavonyl glucoside is not sensitive to glucosidase action at all. Generally, the rate of enzymatic hydrolysis increases as the electron-withdrawing nature of the 4′-substituent increases. An exception is the trifluoromethyl group which, in spite of having the most favourable Hammett constant, does not contribute enough to increase the rate of hydrolysis of its glucoside. The presented experimental results are supported by the electrostatic potential (ESP) analysis and related to the mechanisms of glycoside bond enzymatic hydrolysis.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...