Novel synthetic strategy towards subphthalocyanine-functionalized acetylenic scaffolds via various dibromo-enynes†
Abstract
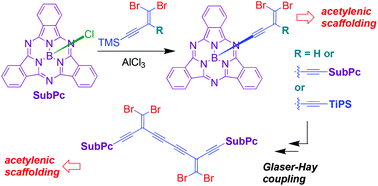
Boron subphthalocyanine (SubPc) is a strong chromophore with interesting applications in the field of functional materials and can be synthetically modified in both the peripheral and axial positions, allowing tuning of optical and redox properties. Herein we present novel acetylenic scaffolds where SubPc units are bridged via acetylenic moieties at the boron atoms. Specifically, we show that dibromo-functionalized enyne and enediyne units (vinylic dibromides) can be attached to one or two SubPc boron atoms using an AlCl3-mediated alkynylation protocol of trimethylsilyl-protected alkynes, and such compounds can conveniently be employed for further Sonogashira or Glaser–Hay coupling reactions. Thereby, new tetraethynylethene (TEE) – SubPc scaffolds were obtained. The degree of communication between two SubPc units incorporated in dimeric scaffolds was investigated by cyclic voltammetry. When bridged by one TEE unit, the oxidations of the SubPc units occurred sequentially, while the two SubPc units behaved as independent redox centers when separated by two TEE units.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...