Preparation of cage-shaped hexakis(spiroborate)s†
Abstract
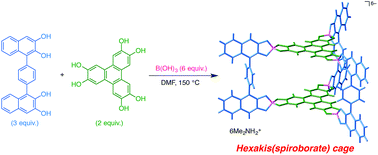
In previous research studies, various types of prismatic cage-shaped molecular containers have been prepared and evaluated in terms of their guest inclusion properties. Basically, most of these molecular cages have a cationic or electron-deficient nature, and exhibit strong affinity mainly toward electron-rich aromatic guests. On the other hand, there is no report concerning anionic prismatic cages that are expected to recognize cationic polyaromatic guests with various structures and functions. In this manuscript, we present the preparation of hexakis(spiroborate)-type molecular cages, which was achieved by the reaction of phenylene- or biphenylenebis(dihydroxynaphthalene), hexahydroxytriphenylene, and boric acid in N,N-dimethylformamide. Their triangular prismatic hollow structures were confirmed by X-ray crystallographic analysis, and it was found that both phenylene- and biphenylene-bridged spiroborate cages have internal cavities of the corresponding size. It was also revealed that tetra(n-butylammonium) cations located inside the cavity and between the two adjacent spiroborate cages resulted in the formation of a one-dimensional columnar array. The molecular recognition behavior of the spiroborate cages was evaluated using tris(pyridinium)triazines as tricationic aromatic guests. 1H NMR measurement implied that a discrete 1 : 1 host–guest complex was formed when 1 equiv. of guest was added to the cage, whereas infinite one-dimensional aromatic stacks were constructed by the addition of 2 equiv. of guest.
- This article is part of the themed collection: Supramolecular chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...