Development of novel C-nucleoside analogues for the formation of antiparallel-type triplex DNA with duplex DNA that includes TA and dUA base pairs†
Abstract
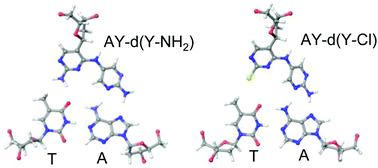
Expansion of the triplex DNA forming sequence is required in the genomic targeting fields. Basically, triplex DNA is formed by the interaction between the triplex-forming oligonucleotides and homo-purine region with the target duplex DNA. The presence of the base pair conversion sites hampers stable triplex formation. To overcome this limitation, it is necessary to develop an artificial nucleic acid to recognize the base conversion sites, and the CG and TA base pairs. We describe the synthesis of C-nucleoside analogues and an evaluation of the ability of triplex formation. Consequently, the combined use of the novel C-nucleoside analogues, AY – AY-d(Y-NH2), AY-d(Y-Cl) and IAP-d(Y-Cl), is capable of recognizing duplex DNA including the TA or dUA base pair.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...