Encapsulation of ionic liquids inside cucurbiturils†
Abstract
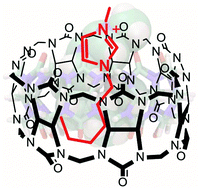
Cucurbit[n]urils (CBn, n = 6–8) serve as molecular receptors for imidazolium-based ionic liquids (ILs) in aqueous solution. The amphiphilic nature of 1-alkyl-3-methylimidazolium guests (Cnmim), with a cationic imidazolium residue and a hydrophobic alkyl chain, enabled their complexation with CBn through a combination of the hydrophobic effect and ion–dipole interactions. 1H NMR experiments revealed that the cavity of CBn can host the hydrophobic chain of the ILs, while one of the carbonyl rims served as a docking site for the imidazolium ring. The structure of the complexes was further analyzed by molecular dynamics (MD) simulations, which indicated that the cavity of CB6 can accommodate up to 5 carbon atoms, while the larger cavity of CB7 and CB8 can encapsulate longer alkyl chains in folded conformations. Isothermal titration calorimetry (ITC) experiments provided up to micromolar affinity of ILs to CBn in aqueous solution, which was independently quantified by indicator displacement titrations.
- This article is part of the themed collection: Supramolecular chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...