Macrocyclization via C–H functionalization: a new paradigm in macrocycle synthesis
Abstract
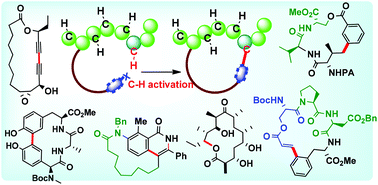
The growing emphasis on macrocycles in engaging difficult therapeutic targets such as protein–protein interactions and GPCRs via preferential adaptation of bioactive and cell penetrating conformations has provided impetus to the search for de novo macrocyclization strategies that are efficient, chemically robust and amenable to diversity creation. An emerging macrocyclization paradigm based on the C–H activation logic, of particular promise in the macrocyclization of complex peptides, has added a new dimension to this pursuit, enabling efficacious access to macrocycles of various sizes and topologies with high atom and step economy. Significant achievements in macrocyclization methodologies and their applications in the synthesis of bioactive natural products and drug-like molecules, employing strategic variations of C–H activation are captured in this review. It is expected that this timely account will foster interest in newer ways of macrocycle construction among practitioners of organic synthesis and chemical biology to advance the field.
- This article is part of the themed collections: Synthetic methodology in OBC and Supramolecular chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...