Synthesis of 1,4-diazepinone derivatives via a domino aza-Michael/SN2 cyclization of 1-azadienes with α-halogenoacetamides†
Abstract
A novel cyclization of α-halogenoacetamides with 1-azadienes has been developed for the efficient preparation of monocyclic 1,4-diazepinones in one step under transition metal-free conditions. Various α-halogenoacetamides and 1-azadienes are well tolerated and give the desired products in good to excellent yields. This cyclization also demonstrates potential synthetic utility on a gram-scale and further transformation.
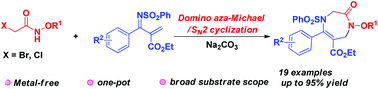


 Please wait while we load your content...
Please wait while we load your content...