Kinetic modelling of acyl glucuronide and glucoside reactivity and development of structure–property relationships†
Abstract
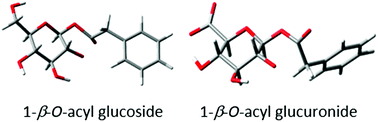
Acyl glucuronide metabolites have been implicated in the toxicity of several carboxylic acid-containing drugs, and the rate of their degradation via intramolecular transacylation and hydrolysis has been associated with the degree of protein adduct formation. Although not yet proven, the formation of protein adducts in vivo – and subsequent downstream effects – has been proposed as a mechanism of toxicity for carboxylic acid-containing xenobiotics capable of forming acyl glucuronides. A structurally-related series of metabolites, the acyl glucosides, have also been shown to undergo similar degradation reactions and consequently the potential to display a similar mode of toxicity. Here we report detailed kinetic models of each transacylation and hydrolysis reaction for a series of phenylacetic acid acyl glucuronides and their analogous acyl glucosides. Differences in reactivity were observed for the individual transacylation steps between the compound series; our findings suggest that the charged carboxylate ion and neutral hydroxyl group in the glucuronide and glucoside conjugates, respectively, are responsible for these differences. The transacylation reaction was modelled using density functional theory and the calculated activation energy for this reaction showed a close correlation with the degradation rate of the 1-β anomer. Comparison of optimised geometries between the two series of conjugates revealed differences in hydrogen bonding which may further explain the differences in reactivity observed. Together, these models may find application in drug discovery for prediction of acyl glucuronide and glucoside metabolite behaviour.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...