Nanocomposite antimicrobials prevent bacterial growth through the enzyme-like activity of Bi-doped cerium dioxide (Ce1−xBixO2−δ)†
Abstract
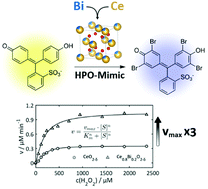
Preventing bacterial adhesion on materials surfaces is an important problem in marine, industrial, medical and environmental fields and a topic of major medical and societal importance. A defense strategy of marine organisms against bacterial colonization relies on the biohalogenation of signaling compounds that interfere with bacterial communication. These reactions are catalyzed by haloperoxidases, a class of metal-dependent enzymes, whose activity can be emulated by ceria nanoparticles. The enzyme-like activity of ceria was enhanced by a factor of 3 through bismuth substitution (Ce1−xBixO2−δ). The solubility of Bi3+ in CeO2 is confined to the range 0 < x < 0.25 under quasi-hydrothermal conditions. The Bi3+ cations are located close to the nanoparticle surface because their ionic radii are larger than those of the tetravalent Ce4+ ions. The synthesis of Ce1−xBixO2−δ (0 < x < 0.25) nanoparticles was upscaled to yields of ∼50 g. The halogenation activity of Ce1−xBixO2−δ was demonstrated with phenol red assays. The maximum activity for x ≈ 0.2 is related to the interplay of the ζ-potential of surface-engineered Ce1−xBixO2−δ nanoparticles and their BET surface area. Ce0.80Bi0.20O1.9 nanoparticles with optimized activity were incorporated in polyethersulfone beads, which are typical constituents of water filter membrane supports. Although Ce1−xBixO2−δ nanoparticles are not bactericidal on their own, naked Ce1−xBixO2−δ nanoparticles and polyethersulfone/Ce1−xBixO2−δ nanocomposites showed a strongly reduced bacterial coverage. We attribute the decreased adhesion of the Gram-negative soil bacterium Pseudomonas aeruginosa and of Phaeobacter gallaeciensis, a primary bacterial colonizer in marine biofilms, to the formation of halogenated signaling compounds. No biocides are needed, H2O2 (formed in daylight) and halide are the only substrates required. The haloperoxidase-like activity of Ce1−xBixO2−δ may be a promising starting point for the development of environmentally friendly, “green” nanocomposites, when the use of conventional biocides is prohibited.


 Please wait while we load your content...
Please wait while we load your content...