A new route for the shape differentiation of cesium lead bromide perovskite nanocrystals with near-unity photoluminescence quantum yield†
Abstract
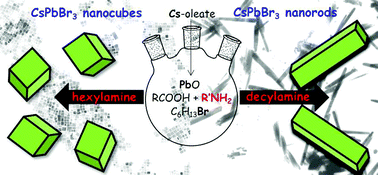
The ongoing interest in all-inorganic cesium lead bromide perovskite nanocrystals (CsPbBr3 NCs) is mainly due to their optical properties, in particular their high photoluminescence quantum yields (PLQYs). Three-precursor synthetic methods, in which the sources of the three elements (cesium, lead and bromine) constituting the perovskite scaffold are chemically independent, often succeed in the achievement of near-unity PLQY perovskite NCs. However, this class of synthetic approaches precludes the accessibility to crystal morphologies different from the traditional cuboidal ones. In order to upgrade three-precursor synthetic schemes to obtain more sophisticated morphologies – such as rods – we propose a conceptually original synthetic methodology, in which a potentially controllable stage of the reaction anticipates the fast crystallization promoted by cesium injection. To this purpose, lead oxide, 1-bromohexane (at different molar ratios with respect to lead) and the ligands (oleic acid and a suitable amine) in 1-octadecene are reacted at 160 °C for an incubation period of 30 min before cesium injection. During this stage and at high C6H13Br/PbO molar ratios, the bromide release from reactions between the ligands and 1-bromohexane promotes the evolution of [PbBr(2+n)]n− species as well as of two-dimensional [(RNH3)2(PbBr4)]n structures with a rod-like shape (aspect ratios ∼10). These structures act as the templating agents for the subsequent crystallization promoted by cesium injection, ensuring the formation of near-unity PLQY nanorods in the presence of decylamine. Conversely, the pronounced decomposition of the preformed [(RNH3)2(PbBr4)]n structures preludes to the formation of near-unity PLQY nanocubes in the presence of hexylamine. The amine choice exerts also an important role in the emission stability of the corresponding NCs, since the nanocubes prepared in the presence of hexylamine maintain their near-unity PLQYs up to 90 days under ambient conditions. In addition to the long-term PLQY stability, the nanorods prepared with decylamine also exhibit a remarkable resistance to the presence of water, due to the compact and hydrophobic organic shell passivating the NC surface. These findings can contribute to the development of innovative synthetic methodologies for controlling the shape and stability of near-unity PLQY perovskite NCs.


 Please wait while we load your content...
Please wait while we load your content...