Synthesizing 1D and 2D metal oxide nanostructures: using metal acetate complexes as building blocks
Abstract
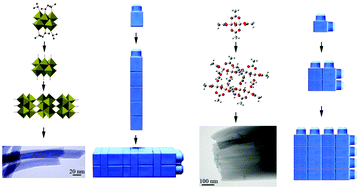
1D and 2D metal oxide nanostructures are important for potential applications in alternative energy, batteries, supercapacitors, catalysts, biomaterials, and electronic nanodevices. Many current approaches for making the desired nanomaterials require multiple steps, which are often exotic and complex for production on a commercial scale. In contrast, the sol–gel reactions between metal alkoxides and organic acids have emerged as a simple protocol for producing metal oxides and inorganic/organic hybrid materials with a controllable 1D or 2D architecture. Our knowledge of this process continues to evolve through the fundamental goal of designing a desired nanostructure from the corresponding molecular building blocks. Our research was driven by the discovery of various morphologies by fine-tuning the synthesis parameters, such as the reaction temperature and molar ratio of the reactants, as well as switching solvents. These discoveries lead to several quesions: What are the building blocks of the 1D and 2D nanostructures and how does the self-assembly occur? What are the reaction kinetics and the mechanisms of nanostructure formation? What role does the solvent play in the assembly process? What are the effects of reaction temperature and pressure? How can we manipulate the nanostructure—for example, the parallel growth of 1D semiconductors—from a substrate surface? And lastly, what are the industrial applications of macroporous aerogels and xerogels? This minireview will highlight documented research accounts to answer these questions.
- This article is part of the themed collection: Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...