Electronic structural regulation of CoP nanorods by the tunable incorporation of oxygen for enhanced electrocatalytic activity during the hydrogen evolution reaction †
Abstract
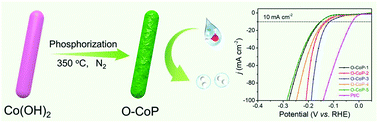
The exploration of cost-effective and highly efficient electrocatalysts for the hydrogen evolution reaction (HER) is of great significance for realizing sustainable H2 production. As previously proposed, anion incorporation in promising earth-abundant transition metal-based electrocatalysts could be a reasonable and competitive approach to regulate the electronic structure with optimized atomic hydrogen adsorption and desorption for enhanced intrinsic electrocatalytic performance during the HER. Herein, we present the rational design and fabrication of O-incorporated CoP (expressed as O-CoP) nanorods with a controllable component and electronic structure. As demonstrated, when the lattice-incorporated O is at an appropriate concentration, the engineered O-CoP nanocatalysts have more active sites exposed with an increased number of electrochemically active areas and better electron/ion conductivity, leading to boosted HER activity and running stability. Typically, the obtained O-CoP nanorods with an optimal oxygen content exhibit excellent HER activity with an overpotential of 116 mV at a current density of 10 mA cm−2 and a small Tafel slope of 59 mV dec−1 in alkaline media. This anion doping strategy may make a widespread contribution to the efficient engineering of electrocatalysts for energy conversion devices.


 Please wait while we load your content...
Please wait while we load your content...