Impact of water on the lubricating properties of hexadecane at the nanoscale†
Abstract
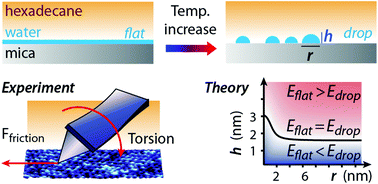
Fluid lubricants are routinely used to reduce friction in a wide range of applications, from car engines to machinery and hard-disk drives. However, their efficiency can be significantly influenced by the ambient conditions they are exposed to, in particular humidity. Our understanding of the molecular mechanisms responsible for the well-documented impact of water on lubrication remains limited, hindering the improvement of tribological formulations. Here, we use Atomic Force Microscopy (AFM) and shear force spectroscopy to investigate the structural and dynamical behaviour of a model lubricant, hexadecane, confined between an AFM probe and a hydrophilic mica surface at different temperatures and humidities. We show that both the nanoscale structure and the tribological behaviour of the system are dominated by the nucleation of water nanodroplets at the interface. The process is favoured at higher temperature and can be explained with classical nucleation theory whereby the droplets become stable when larger than 20 nm to 50 nm size, depending on the ambient conditions. Below this threshold, a molecularly thin film of water molecules coats the surface uniformly. Highly localised shear measurements demonstrate a detrimental impact of the nanodroplets on shear with a twofold increase in the lubricated friction force. However, this can be mitigated by the adjunction of an amphiphilic additive, here oleic acid.


 Please wait while we load your content...
Please wait while we load your content...