Directional Diels–Alder cycloadditions of isoelectronic graphene and hexagonal boron nitride in oriented external electric fields: reaction axis rule vs. polarization axis rule†
Abstract
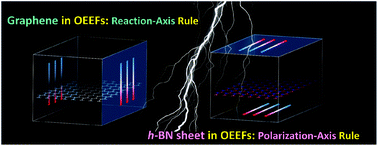
The present study introduces the mechanisms for the oriented external electric field (OEEF)-participating cycloadditions of nanographene and the analogous hexagonal boron nitride (h-BN) nanoflakes. Despite the C–C and B–N pairs being isoelectronic, their different ionicities give rise to their distinct response to applied electric fields. For the nanographene models, the Diels–Alder addition obeyed the reaction axis rule and the activation barrier changed under an OEEF perpendicular to the carbon skeleton for enhanced/reduced intermolecular charge transfer, which provides a feasible strategy for the side-selective derivatization of graphene to obtain one-face-only adducts and Janus bifunctional products. By contrast, for the h-BN models, the variation of the activation barrier was pronounced when the electric field was aligned along the in-plane N–B bond rather than the well-accepted reaction axis. Electronic structure analyses indicated that, because of the opposite electron withdrawing/donating nature of the reacting sites of B/N, an OEEF along the N–B bond was capable of further enhancing the polarization via in-plane intramolecular charge transfer, resulting in a stabilized transition state and notable barrier reduction.


 Please wait while we load your content...
Please wait while we load your content...