Ni or FeO nanocrystal-integrated hollow (solid) N-doped carbon nanospheres: preparation, characterization and electrochemical properties†‡
Abstract
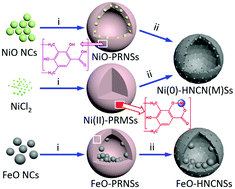
In this paper, phase-pure monodisperse NiO nanocrystals were prepared in a temperature-dependent manner via a thermal decomposition approach, showing sphere-like shapes and snowflake-like NiO arrays. Such hydrophobic NiO nanocrystals were converted into hydrophilic nickel oxide-sodium oleate-Pluronic P123 (NiO-SO-P123) micelles in aqueous solution. Phenolic resin (PR) formed in situ was successfully deposited on the hydrophilic area of the NiO-SO-P123 micelles via a heterogeneous nucleation mechanism to form NiO-phenolic resin nanospheres (NiO-PRNSs) with uniform particle size. By adjusting the size and amount of NiO nanocrystals used, the diameter of the obtained NiO-PRNSs can be effectively controlled from 185 to 103 nm, and a narrow size distribution is seen, revealing the effects of the NiO nanocrystals on the reconstructed NiO-integrated micellar size. Meanwhile, the morphology (ring buoy, semi-bowl, sphere) depends upon the initial amount of NiO. The carbonization of NiO-PRNSs produced Ni(0)-integrated hollow N-doped carbon nanospheres (Ni(0)-HNCNSs), which involved the conversion of NiO to Ni(0) and the contraction of particle size, and the size and distribution was affected by the starting amount of NiO. However, upon using monodisperse and polyhedral FeO nanocrystals, the obtained FeO-free/-incompletely-filled/-fully-filled core–shell structured Fe-PRNSs showed relatively uniform particle size, except for when multiple FeO cores formed large FeO-PR nanospheres after starting with the same initial FeO size. The carbonized FeO-HNCNSs still preserved a pomegranate-like core–shell structure with uniform size and there was no change in the size of the FeO nanocrystals. Moreover, high-loaded Ni(0)-integrated hollow or solid N-doped carbon microspheres or flakes can be synthesized via a one-pot method, but with a broad size range, showing highly uniform Ni distribution with a Ni size as small as 8.5 nm. Note that Ni(0)- and FeO-HNCNSs were prepared for the first time according to our knowledge. Finally, low-loaded Ni- and FeO-HNCNSs with uniform morphology and size were chosen as representatives to investigate their electrochemical properties for lithium-ion batteries (LIBs), showing excellent lithium storage properties and superior reversibility. This study provides a potential strategy for controlling the sizes and morphologies of metal-integrated carbon materials to obtain adjustable electrochemical properties.
- This article is part of the themed collection: Celebrating 60 years of the Fujian Institute of Research on the Structure of Matter


 Please wait while we load your content...
Please wait while we load your content...