Enhancing photocatalytic H2 evolution on In2S3/mesoporous TiO2 nanocomposites via one-pot microwave-assisted synthesis using an ionic liquid†
Abstract
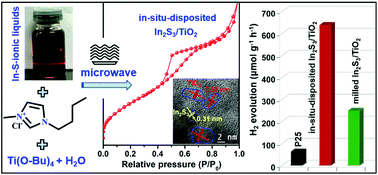
Charge transfer and surface photocatalytic reactions are two crucial factors that determine the efficiency of photocatalysts in converting light energy to hydrogen energy. Herein, a fast and green strategy composed of a one-pot microwave-assisted solvothermal synthesis followed by a heat treatment process is developed for synthesizing In2S3/mesoporous TiO2 nanocomposites, using a mixture of tetra-butyl titanate, H2O, In-S-ionic liquid precursor solution (In-S-ILs) and [Bmim]Cl as the reactants. The ionic liquid cations act not only as a microwave absorbent during the solvothermal process, but also as a morphology-controlling agent via a dissolution-recrystallization process, leading to the formation of nanocomposites consisting of small and uniform sized In2S3 and mesoporous TiO2. The optimum In2S3/mesoporous TiO2 nanocomposite with an In2S3 content of 1.01 mol% exhibits a H2 evolution rate of 637.9 μmol g−1 h−1, which is over 10 times that of commercial P25, and is 5.7 and 2.6 times that of neat mesoporous TiO2 and the milled In2S3/mesoporous TiO2 with the same component, respectively. The improved H2 evolution activity is predominantly attributed to the small size of In2S3, special mesoporous structure of TiO2, and their intimate heterojunction interface, which guaranteed effective charger transfer between In2S3 and mesoporous TiO2 as well as abundant active sites for photocatalytic reactions. This work provides a novel and simple strategy for obtaining special structures of semiconductor-based nanocomposites for efficient energy conversion.
- This article is part of the themed collection: Celebrating 60 years of the Fujian Institute of Research on the Structure of Matter


 Please wait while we load your content...
Please wait while we load your content...