Electroactivation-induced IrNi nanoparticles under different pH conditions for neutral water oxidation†
Abstract
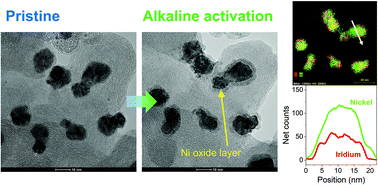
Electrochemical oxidation processes can affect the electronic structure and activate the catalytic performance of precious-metal and transition-metal based catalysts for the oxygen evolution reaction (OER). Also there are emerging requirements to develop OER electrocatalysts under various pH conditions in order to couple with different reduction reactions. Herein, we studied the effect of pH on the electroactivation of IrNi alloy nanoparticles supported on carbon (IrNi/C) and evaluated the electrocatalytic activities of the activated IrNiOx/C for water oxidation under neutral conditions. In addition, their electronic structures and atomic arrangement were analyzed by in situ/operando X-ray absorption spectroscopy (XAS) and identical location transmission electron microscopy techniques, showing the reconstruction of the metal elements during electroactivation due to their different stabilities depending on the electrolyte pH. IrNiOx/C activated under neutral pH conditions showed a mildly oxidized thin IrOx shell. Meanwhile, IrNiOx/C activated in acidic and alkaline electrolytes showed Ni-leached IrOx and Ni-rich IrNiOx surfaces, respectively. Particularly, the surface of IrNiOx/C activated under alkaline conditions shows IrOx with a high d-band hole and NiOx with a high oxidation state leading to excellent OER catalytic activity in neutral media (η = 384 mV at 10 mA cm−2) whereas much lower OER activity was reported under alkaline or acid conditions. Our results, which showed that electrochemically activated catalysts under different pH conditions exhibit a unique electronic structure by modifying the initial alloy catalyst, can be applied for the design of catalysts suitable for various electrochemical reactions.
- This article is part of the themed collection: Advanced Nanomaterials for Energy Conversion and Storage


 Please wait while we load your content...
Please wait while we load your content...