Electrochemical intercalations of divalent ions inside Ni/Zn co-doped cobalt sulfide nanoparticle decorated carbon spheres with superior capacity†
Abstract
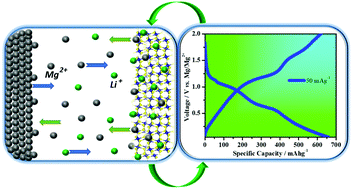
Among post-lithium ion batteries, magnesium ion batteries (MIBs) are receiving growing attention due to their divalent nature, intrinsic low cost, dendrite free cycling, and atmospheric stability. However, their realization is constrained because of the absence of suitable cathodes that can accommodate Mg2+ with fast reversibility. To bypass the sluggish movement of Mg2+ ions inside the cathode and utilize the full advantage of the Mg anode, a Mg2+/Li+ hybrid ion battery (MLIB) is introduced here with rationally designed porous Ni/Zn co-doped CoS2@C spheres as the cathode material. The Ni/Zn-CoS2@C cathode with high porosity and electrical conductivity showed an appreciable specific capacity of 158 mA h g−1 at 20 mA g−1 for MIBs, which was significantly boosted up to 667 mA h g−1 at a current density of 50 mA g−1 by employing Mg2+/Li+ hybrid electrolytes. Their specific capacity and the corresponding energy density (614 W h Kg−1) are the highest among MLIBs and comparable to those of lithium ion batteries. Furthermore, MLIBs displayed significant cycling stability by retaining the maximum specific capacities of 324.6 and 230 mA h g−1 at 100 and 500 mA g−1, respectively after 100 cycles. The excellent electrochemical properties of the synthesized cathodes are attributed to their high porosities and electrical conductivities, the synergistic effect of doped species and their capability to accommodate both Mg2+ and Li+ ions without side reactions. Various ex situ characterization tools were employed to develop further understanding of the intercalation chemistries and mechanisms of both Mg2+ and Li+ ions inside host materials.


 Please wait while we load your content...
Please wait while we load your content...