Interstratified heterostructures of metal hydroxide nanoclusters and MoS2 monolayers with improved electrode performance†
Abstract
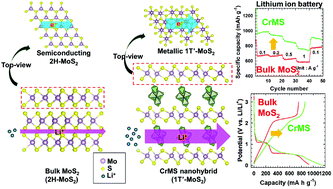
Interstratified 2D nanohybrids of chromium hydroxide–molybdenum disulfide with improved electrode functionality are synthesized by the self-assembly of anionic monolayered MoS2 nanosheets with cationic chromium hydroxide nanoclusters. The intercalative hybridization of MoS2 with chromium hydroxide nanoclusters leads to a significant increase of basal spacing as well as to the formation of an open porous stacking structure. This is the first example of metal hydroxide nanocluster-pillared transition metal dichalcogenide (TMD) hybrid materials. According to extended X-ray absorption fine structure analysis, open tetrameric chromium hydroxide nanoclusters are stabilized in-between metallic 1T′-MoS2 monolayers. In comparison with the pristine MoS2 material, the chromium hydroxide-pillared molybdenum disulfide nanohybrids show remarkably improved charge storage capacity with excellent rate performance for lithium ion batteries, highlighting the beneficial effect of pillaring with metal hydroxides on the electrode performance of MoS2. The improvement of electrode functionality upon hybridization is attributable to the increase of basal spacing, the stabilization of metallic 1T′-MoS2 content, the improvement of charge transfer kinetics, and the stabilization of the open porous structure upon electrochemical cycling. The present study clearly demonstrates that an electrostatically-driven self-assembly between exfoliated TMD nanosheets and cationic inorganic nanoclusters can provide an effective way of synthesizing heterostructured hybrid electrode materials with improved performance.
- This article is part of the themed collections: Advanced Nanomaterials for Energy Conversion and Storage and Editor’s Choice: Single-atom and nanocluster catalysis


 Please wait while we load your content...
Please wait while we load your content...