Targeted GSH-exhausting and hydroxyl radical self-producing manganese–silica nanomissiles for MRI guided ferroptotic cancer therapy†
Abstract
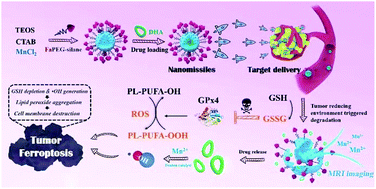
Ferroptosis, a cell death path induced by the generation of reactive oxygen species (ROS), will cause the accumulation of lipid peroxides (PL-PUFA-OOH) and achieve potent tumor-regression. However, glutathione (GSH)-dependent glutathione peroxidase 4 (GPx4) can reduce PL-PUFA-OOH and antagonize the ferroptosis inducing effect of ROS. Herein, folate-PEG modified dihydroartemisinin (DHA) loaded manganese doped mesoporous silica nanoparticles (described as nanomissiles) were constructed for integrating the effect of GSH exhaustion and ROS generation. After endocytosis by tumor cells, intracellular GSH triggered the degradation of nanomissiles, which allowed the simultaneous release of DHA and Fenton catalytic Mn2+ due to the redox reaction between the manganese–oxygen bonds and GSH. The degradation would lead to GSH exhaustion, activation of Mn2+-based magnetic resonance imaging (MRI), and DHA-driven ˙OH generation. The GSH-free environment inhibited the activity of GPx4 and enhanced the accumulation of PL-PUFA-OOH oxidized by ˙OH. Furthermore, the cooperative effects suppressed tumor metastasis by destroying the structure of polyunsaturated fatty acids in the cell membranes and showed potent antitumor activity. This innovative ferroptotic therapy integrating the GSH exhaustion and ROS generation will be a promising strategy for cancer therapy.


 Please wait while we load your content...
Please wait while we load your content...