Theoretical anchoring effect of new phosphorus allotropes for lithium–sulfur batteries†
Abstract
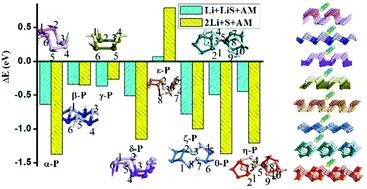
The lithium–sulfur battery system has appeared as a new-generation alternative to lithium-ion batteries with 5–7 times higher specific energy density than conventional lithium-ion batteries. But its commercial implementation is still impeded by a series of technical challenges. Modifying separation with the anchoring material is viewed as an effective way to overcome these problems and achieve long-term cycling stability and high-rate performance. In this paper, eight phosphorus allotropes, α-P, β-P, γ-P, δ-P, ε-P, ζ-P, θ-P and η-P, are investigated as anchoring materials for separation in lithium–sulfur batteries based on binding energy and diffusion barrier discussion. The results show that the crystal structures of phosphorus polymorphs are crucial to the suitability of their application as anchoring materials for lithium–sulfur batteries. The binding energy results show that except ε-P, all the other phosphorus allotropes can strike a balance between binding strength and intactness of the Li2Sn species. Among these, ε-P composed of P4 squares is a strong anchoring material and may lead to the decomposition of the Li2S cluster. Furthermore, for Li atoms and lithium sulfides, the most convenient diffusion paths are all along the grooves in the phosphorus monolayers, including the life-boat shaped (α-P, γ-P, δ-P and ε-P), wave shaped (β-P, ζ-P) and back-to-back shaped (θ-P, η-P) grooves. Except ε-P, all the other phosphorus allotropes can be applied to modify the separation for lithium–sulfur batteries. But among these, α-P, β-P, γ-P and δ-P composed of P6 hexagons can act as more suitable substrates for Li atom and lithium sulfide diffusion with lower barriers. As a result, α-P, β-P, γ-P and δ-P composed of P6 hexagons are good choices as separation modification materials in lithium–sulfur batteries among phosphorus allotropes. In addition, the structure correlations among α-P, β-P, γ-P and δ-P, and the likely conversion pathways from α-P to β-P, from β-P to γ-P, and from γ-P to δ-P are discussed.


 Please wait while we load your content...
Please wait while we load your content...