A direct Z-scheme PtS2/arsenene van der Waals heterostructure with high photocatalytic water splitting efficiency†
Abstract
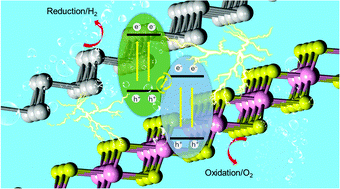
To overcome current serious energy and environmental issues, photocatalytic water splitting holds great promise because it requires only solar energy as an energy input to produce hydrogen. In this work, based on first-principle calculations, we studied the van der Waals heterostructure formed by PtS2 and arsenene (Are) monolayers that were successfully synthesized on a large scale at high quality. From an analysis of the migration paths of photoinduced electrons and holes, a direct Z-scheme photocatalytic mechanism is demonstrated in this heterostructure. Furthermore, the PtS2/Are direct Z-scheme heterostructure has decent band edge positions to promote the redox reaction to decompose water at pH 0. The interfacial charge difference and potential drop are presented, which further support the formation of a direct Z-scheme photocatalyst. More importantly, the PtS2/Are heterostructure has quite high solar-to-hydrogen (STH) efficiency (49.32%), significantly enhanced compared with isolated PtS2 (12.67%) or Are (10.34%) monolayers. This direct Z-scheme PtS2/Are heterostructure with excellent STH efficiency suggests its promising application as a photocatalyst for water splitting.
- This article is part of the themed collection: Nanoscale 2021 Lunar New Year Collection

 Please wait while we load your content...
Please wait while we load your content...