Pt nanoparticle-decorated two-dimensional oxygen-deficient TiO2 nanosheets as an efficient and stable electrocatalyst for the hydrogen evolution reaction†
Abstract
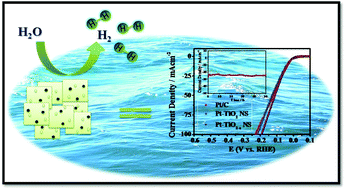
Developing novel hydrogen evolution reaction (HER) catalysts with high activity, high stability and low cost is of great importance for the ever-broader applications of hydrogen energy. Among the conventionally used platinum-based heterogeneous catalysts, the high consumption and low utilization efficiency of precious platinum are the most crucial issues. Herein we present a facile approach to prepare an effective HER catalyst with platinum nanoparticles dispersed on oxygen-deficient TiO2−x nanosheets (NSs). The fabricated Pt–TiO2−x NS electrocatalyst shows an overpotential of 35 mV at 10 mA cm−2 for the HER in 0.5 M H2SO4, which is highly comparable to that of commercial Pt/C (34 mV). More attractively, the Pt–TiO2−x NS electrocatalyst largely enhanced the mass activity (MA) of Pt and electrochemical stability compared to commercial Pt/C. The excellent HER performance of Pt–TiO2−x NSs is attributed to the synergetic effect between highly dispersed Pt species and TiO2−x NSs with oxygen vacancies, which enhances both electrocatalytic activity and durability over a wide pH range. This strategy can provide insights into constructing highly efficient catalysts and their support for different energy-related applications.


 Please wait while we load your content...
Please wait while we load your content...